I’ve been on a bit of a health kick since moving to SF about eight months ago. At first, I still carried over a few of my college habits: binge drinking, smoking, and eating semi-healthy without really tracking macros or calories. Compared to college, I was doing much better: eating out only two or three times a week, maybe smoking a cigarette on a night out (still terrible for you).
But deep down, as my workload grew and my weekday hours became increasingly scarce, I knew I had to take a harder look at my habits, to create systems that actually optimized for brain health, not just for superficial reasons, but for clarity, creativity, and longevity.
That’s why attending the Brain Optimization Summit, hosted by MBC Biolabs during SF Tech Week, felt perfectly timed: a convergence of my personal health journey and my fascination with neurobiology and longevity science.
Longevity seems to be all the rage right now in Silicon Valley, with the rise of health-focused influencers like Bryan Johnson, Peter Attia, and Andrew Huberman perpetuating conversations on the latest breakthroughs in extending healthspan—and translating those findings into actionable insights for the average person. Within these discussions, there’s a growing emphasis on brain health, underscoring how longevity and the mind are deeply intertwined. After all, the longevity movement isn’t just about extending life; it’s about improving the quality of those years.

What’s the point of living longer if your bodily mechanisms still reflect those of an elderly individual facing age-related neurodegenerative disease?
The discussions at the Brain Optimization Summit were thought-provoking, from using AI platforms to derive brain organoid models for wet-lab experimentation to exploring novel drug candidates aimed at treating rare neurological diseases in children.
Here are some of the biggest takeaways and insights into the future of brain health and its implications for longevity.
Across talks from CEOs, CSOs, clinicians, and more across the field, certain stats echoed:
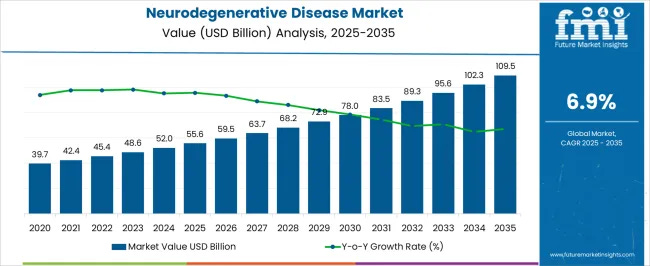
Both market and moral incentives are driving this expansion, fueled by an aging population and a surge in AI-driven biopharma innovation.
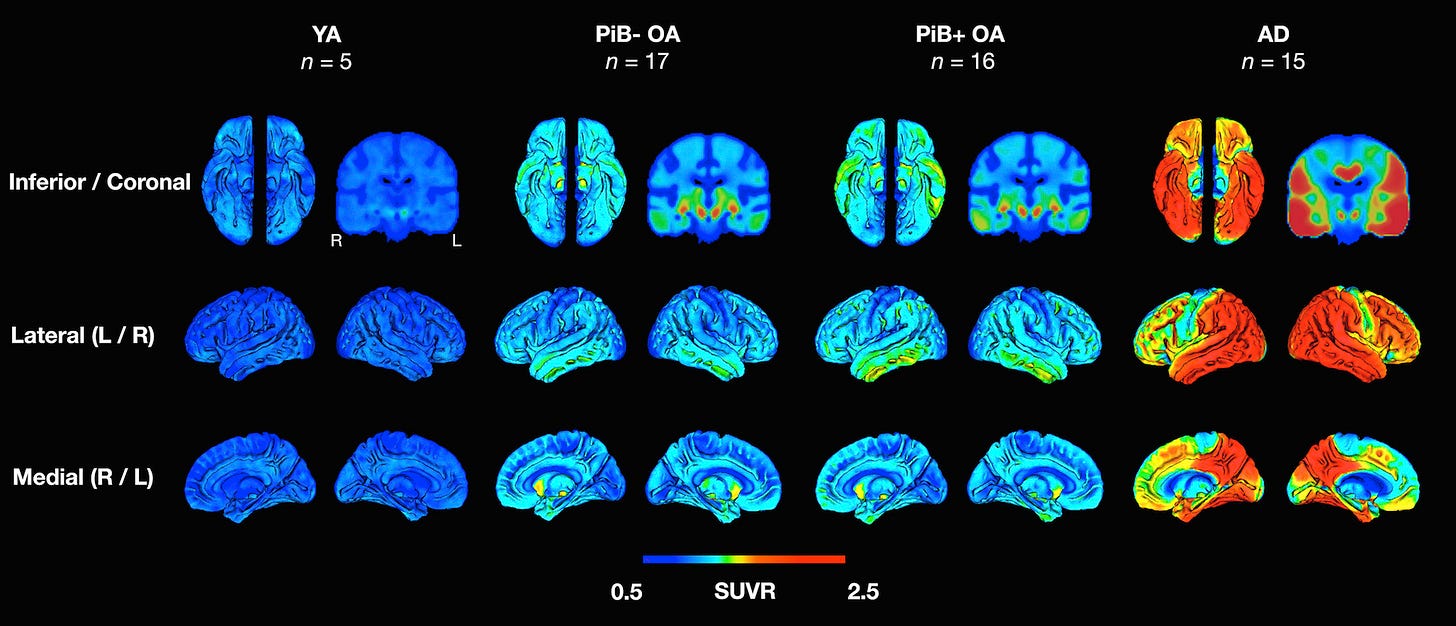
Sitting in on a talk by Dr. Christine Glorioso, CEO and co-founder of NeuroAge Therapeutics, was eye-opening to say the least. Her company focuses on developing drugs for neurodegenerative diseases like dementia and Alzheimer’s, conditions that sit at the crossroads of aging and cognition.
One particularly striking slide showed a series of PET scans across different age groups, mapping the accumulation of amyloid-β (Aβ) and tau proteins, hallmarks of neural decline. You could literally see the buildup intensify with age.
According to the World Health Organization (WHO), the global population aged 60 and above is expected to double from 1.2 billion in 2020 to nearly 2.4 billion by 2050. Even more striking, the number of individuals aged 80 and above will triple, reaching around 426 million.
These numbers are staggering, and as people live longer, there’s an urgent need to not only address age-related diseases but also to understand the biological mechanisms that underlie longevity itself.
This is, in many ways, the essence of the longevity movement. If researchers and entrepreneurs can better understand the brain—an organ that orchestrates nearly every biological process in the body—they may uncover deeper insights into what it actually means to live longer. Because at its core, longevity isn’t just about extending life; it’s about preserving clarity, vitality, and quality of life along the way.
It’s not exactly groundbreaking to hear that lifestyle plays a role in brain health, but it’s one of those truths that’s easy to overlook. The biggest contributors?
Diet. Exercise. Sleep.
Even minor lifestyle changes can yield significant benefits.
At the summit, Dr. Glorioso shared the story of Simon Nicholls, a Florida man carrying two copies of the APOE4 gene, one of the strongest genetic predictors for Alzheimer’s. Despite his predisposition, Nicholls managed to nearly halve his risk of developing the disease by undergoing extensive lifestyle changes: diet adjustments, reducing stress, improving sleep, and exercising regularly.
Running, in particular, emerged as a recurring theme across multiple speaker sessions in regards to its cognitive and healthspan benefits. Running has been shown to reduce cognitive decline, lower the risk of neurodegeneration, and be associated with overall longer lifespans. A 2017 New York Times article even suggested that running just one hour per week can add 7 hours to your life. Imagine that!

When it comes to diet, the Mediterranean Diet (MD) remains a gold standard for promoting both brain and heart health. Characterized by its low protein, Omega-3 rich, high vegetable and fruit content attributes, the MD has been shown to reduce inflammatory biomarkers, which are central to many age-related metabolic and cardiovascular diseases.
Other factors, like stress management, mental stimulation, and minimizing microplastic exposure, also came up throughout Dr. Glorioso’s talk. But the underlying message was clear: sustainable habits compound. Maintaining healthy lifestyle practices isn’t just about optimizing for brain health or longevity in isolation: it’s about supporting the entire ecosystem of the body, so that all of it can age better, together.
Because while therapeutics are advancing, the simplest interventions, lifestyle, diet, and movement, remain the first line of defense against the very diseases we’re working so hard to treat.
The blood–brain barrier (BBB) is one of the biggest challenges in developing neurotherapeutics. It’s a specialized cellular membrane that tightly regulates what crosses from circulating blood into the brain. Composed of endothelial cells (ECs) lining blood vessel walls, the BBB is a fortress for the central nervous system.
The catch?
The BBB blocks nearly 98% of small-molecule drugs from entering the brain. That’s why developing effective neurotherapies has been so difficult for decades, and also emerging innovations in this space so exciting.
One of the summit speakers, the CEO and founder of Concordance Medical, showcased a novel FDA-designated fast-track technology that uses low-level ultrasound waves to gently open the BBB for about 90 minutes, long enough to allow therapies to pass through and reach the brain effectively. CEO of Adjuvia Therapeutics, spoke of their molecular approach in addressing the BBB issue: using a nano-encapsulation formula to enable entry into the brain.
As an engineer, I was fascinated by these discussions surrounding the BBB. I’m trained to think in systems, to find the most efficient path through complexity. And it’s clear that with both hardware and molecular approaches, the BBB is simply another system to hack: impossibly intricate, but still governed by logic and constraints that can, eventually, be solved.
Some neurodegenerative diseases exhibit sex-based patterns, with some disproportionately impacting women, and others disproportionately affecting men. These differences point to an urgent need for therapies and biomarker-driven, multi-omics approaches that account for sex as a biological variable when stratifying patients in clinical trials.
Dr. Laura Glickman, CEO and co-founder of Adjuvia Therapeutics, discussed the company’s lead asset, ATI-103, an oral astaxanthin nanoparticle designed to treat Friedreich’s ataxia (FA)—a rare neurodegenerative disease that causes loss of coordination and movement. While FA itself affects both sexes, understanding how mitochondrial and hormonal factors contribute to disease progression could help tailor future treatments, including those built on platforms like ATI-103.
ATI-103 stands out for its ability to deliver astaxanthin, a metabolic stabilizer, antioxidant, anti-inflammatory, and mitochondrial protector, through a nano-encapsulation formula that enables entry into both the brain and heart, two organs heavily impacted in FA and in other neurodegenerative conditions that exhibit sex-linked vulnerability.
Many of these diseases share overlapping mechanisms, particularly mitochondrial dysfunction and oxidative stress. Research increasingly suggests that hormonal shifts, especially the decline in estrogen during menopause, can heighten mitochondrial vulnerability and oxidative damage in the brain, heart, and ovaries. This may help explain why women face a higher risk for certain neurodegenerative conditions, such as Alzheimer’s and multiple sclerosis.
That’s why compounds like astaxanthin are interesting. Early studies indicate it may enhance mitochondrial efficiency and reduce oxidative stress, positioning it as a potential bridge between neuroprotection, hormonal health, and longevity. Exploring these mechanisms through a sex-specific lens could ultimately bring us closer to more personalized, equitable therapies for neurodegenerative disease.
As an engineer, I found this summit inspiring for so many reasons. But in a place like Silicon Valley, obsessed with systems, optimization, and hacking, it’s fascinating to imagine what it means to hack the original system: the human brain.